The seventeen year old patient had a painful, rapidly growing lump on the back of her right hand. She had pinched the unlucky appendage between two railway carriage seats on a transcontinental trip to Alaska some months before, and when the bruise failed to heal she assumed the injury had become infected. However the bruise turned into a bulge, the pain steadily worsened, and her baffled doctors were eventually compelled to call for Dr. Coley. As a surgical man, Coley would never have guessed that this innocuous referral would take his career in a totally new direction– into an unusual branch of medicine now known as cancer immunotherapy.
At first Dr. Coley was also uncertain about the diagnosis. But as the girl’s condition rapidly deteriorated– with the lump becoming larger, more painful, and associated with the loss of sensation in some of the surrounding skin– the awful truth became apparent. She had a sarcoma, a type of cancer that affects bone and connective tissue in the body. Unfortunately, 19th century medicine offered very few treatment options.
On November 8th, Coley amputated her arm at the elbow. Although the operation appeared to go well, the girl– named Elizabeth Dashiell– developed severe abdominal pain three weeks later. Soon thereafter she noticed more lumps in her breasts and armpits, signs that the cancer was metastasizing, or spreading. She rapidly lost strength and died on January 23rd 1891, a scant three and a half months after her initial consultation, with a traumatized Dr. Coley at her bedside.
Elizabeth’s death hit the young surgeon hard. While a more experienced physician might have shrugged away the apparent failure and moved on, Coley was determined to do something. His ensuing efforts culminated in the development of a famous fluid that, for a time, appeared to promise the fulfillment of that long-held dream: a universal cure for cancer.
Coley began by poring through the hospital’s records, looking for clues from previous sarcoma cases that might lead to better treatments in the future. He soon found what he was looking for: the case of a German man who came to the hospital with an egg-sized sarcoma in his left cheek some seven years earlier. There were several attempts to excise the tumour but none of them were successful– each time the cancer came back, as aggressive as before. The final operation could only partially remove the huge mass, leaving an open wound that subsequently became infected.  William B. ColeyThe unfortunate immigrant was deemed a terminal case.
William B. ColeyThe unfortunate immigrant was deemed a terminal case.
Yet four and a half months later, the man was discharged with no trace of disease. Coley personally tracked down the former patient to verify that the miraculous cure had taken place. Indeed, the man was healthy and happily settled into his new life in the United States. The records showed that after the wound became infected with a commonplace bacterium, Streptococcus pyogenes, the patient went through several bouts of fever. With each attack of fever the tumour shrank until eventually it disappeared entirely, leaving only a large scar under the left ear. Coley surmised that the infection had stimulated the German’s immune system– as evidenced by the repeated fevers– and that it was this immune response that had caused the eradication of the cancer.
The story so convinced Coley that he– perhaps cavalierly– contrived to contaminate his next ten suitable sarcoma cases with Streptococcus. His initial approach was to inject a solution of live bacteria deep into the tumour mass on a repeated basis over several months. The first patient to undergo this treatment was a bedridden man with inoperable sarcoma in the abdominal wall, bladder, and pelvis. Using this experimental method, the patient was cured spectacularly. He staged a full recovery, and survived another twenty-six years before dying from a heart attack. But subsequent results were mixed; sometimes it was difficult to get the infection to take hold, and in two cases the cancer responded well to treatment but the patients died from the Streptococcus infection.
Coley’s discovery, as it turns out, was actually a re-discovery. The idea of a link between acute infection and the resolution of tumours was not new, and the phenomenon of infection-related “spontaneous regression” of cancer has been documented throughout history. A 13th century Italian saint was reputed to have his tumour-afflicted leg miraculously healed shortly after the malignant growth burst through the skin and became infected. Crude cancer immunotherapies working along similar lines to Coley’s early experiments were known in the 18th and 19th centuries, and may extend back to the time of the pharaohs. Ancient writings suggest that the renowned Egyptian physician Imhotep may have used a similar infect-and-incise method to treat tumours.
But Coley took those first important steps in dragging this old remedy into the twentieth century. After the fatalities with the ‘live’ version of his therapy, he developed an improved fluid containing killed bacteria of two different strains, Streptococcus pyogenes and Serratia marcescens. This was based on the idea that the dead bacteria would still have the immune-stimulating capability of their living brethren (in the form of purported ‘toxins’), but not share their inconvenient tendency to cause death. 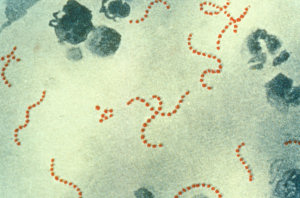 Streptococcus pyogenesHis invention became variously known as ‘Coley’s Toxins’, ‘Coley’s Vaccine’, ‘Mixed Bacterial Toxins’ or ‘Coley Fluid.’ The treatment was met with considerable success, with one study in 1999 suggesting that it was at least equally as effective in treating cancer as conventional modern therapies. With due care in dosing and management of the induced fever, it was also remarkably safe.
Streptococcus pyogenesHis invention became variously known as ‘Coley’s Toxins’, ‘Coley’s Vaccine’, ‘Mixed Bacterial Toxins’ or ‘Coley Fluid.’ The treatment was met with considerable success, with one study in 1999 suggesting that it was at least equally as effective in treating cancer as conventional modern therapies. With due care in dosing and management of the induced fever, it was also remarkably safe.
Although Coley took the concept of immunotherapy much further than his pharaonic forebears, he had no clear idea how his toxins actually worked, and the tools did not yet exist for him to find out. But given the rapid scientific progress at the turn of the last century, he reasoned that a deeper understanding of his therapy would arrive soon enough. Although the true extent of his “Toxin” success has been questioned by historians, the validity of his approach has never been seriously called into doubt. Indeed his results are regularly cited in the cancer research literature to this day.
Over the following years Coley continued to refine his technique. He determined that the toxins should be administered to patients at progressively higher doses to counter the body’s innate “immune tolerance” to the treatment. Other physicians in America and Europe also experimented with the method, and found that the toxins appeared to work just as well in a number of different non-sarcoma cancer types such as carcinoma, lymphoma, and melanoma. They could also be given intravenously some distance from the site of the tumour, and still be effective. Variations on the basic bacterial recipe and different dosing regimes were tried, depending on the individual patient and the particular cancer’s type and proliferation. Through his career Coley himself treated over one hundred patients with his concoction, and countless more were treated by other doctors.
As the fame of his fluid grew, so did Dr Coley’s stature: in 1915 he became head of the Bone Service at the New York Memorial Hospital (which later became the Memorial Sloan-Kettering Cancer Center). By the time he died in 1936, Coley’s Toxins were mentioned in a number of different surgical textbooks as a standard anti-cancer therapy.
Conventional modern medicine, however, very rarely employs Coley’s Toxins in the treatment of cancer, for reasons almost as complicated as the human immune system itself. One concern is the far-from-complete understanding of the mechanism of action; generally, doctors are reluctant to administer treatments whose workings they don’t fully comprehend. The stimulated human immune system is a whirling tempest of different physiological and biochemical responses, and even now there’s much uncertainty about how Coley’s Toxins modified this complex mechanism to better attack its cancerous target. 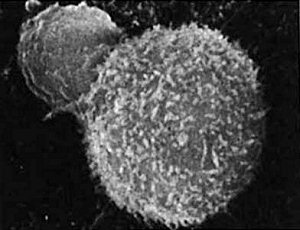 T-cell (small sphere) attacks a cancer cellOne theory stresses the importance of the fluid-induced fever in killing the cancer cells; another considers the debris-engulfing macrophage cells to be the main players, while others consider various different immune messenger molecules– or cytokines– to be important.
T-cell (small sphere) attacks a cancer cellOne theory stresses the importance of the fluid-induced fever in killing the cancer cells; another considers the debris-engulfing macrophage cells to be the main players, while others consider various different immune messenger molecules– or cytokines– to be important.
The eclipsing of Coley’s Toxins also had something to do with the concurrent development of radiation therapy and, a little later, chemotherapy. Soon after Wilhelm Roentgen discovered X-rays in 1895, the possibility of using radioactivity to treat cancer was investigated. The technology was exciting, new, and developing fast along well-understood principles. Although the first results of radiotherapy weren’t all that impressive, it had the advantage of fractional doses, and once the equipment was in place it didn’t require the complicated, patient-specific preparation which was needed with Coley’s Toxins. Likewise chemotherapy was based on known scientific principles, and could be manufactured and used relatively easily.
Furthermore, both radiotherapy and chemotherapy have an immune-suppressing side-effect. Since both treatments kill the rapidly dividing cells of the immune system along with the rapidly dividing cancer cells, both can be used together if care is taken. But immune-stimulating Coley’s Toxins work entirely differently, and their effect would be cancelled out if used at the same time as high-dose immunosuppressant chemo- or radiotherapy. It became an either/or situation– and in the end, the fashionable new treatments won out over Coley’s fiddly reworking of an ancient ‘natural’ remedy.
So when the US Food and Drug Administration changed the status of Coley’s Toxins to that of a ‘new drug’ in 1963– meaning that it could only be used in clinical trials, and greatly reducing its availability– it seemed that its time had already long passed. But cancer immunotherapy does have limited applications today. Perhaps its most frequent mainstream use is in the treatment of bladder cancer; solutions containing the tuberculosis vaccine are routinely instilled into cancer-affected bladders, and are effective in causing regression of tumour deposits. It is theorized that the bladder’s immune response deals with the cancer in a similar way to the whole-body immune effect of Coley’s Toxins. 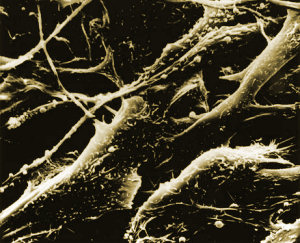 Melanoma cellsMelanoma, a particularly nasty type of skin cancer that responds poorly to conventional radiotherapy and chemotherapy, is sometimes treated with an immune-stimulating cytokine called interferon.
Melanoma cellsMelanoma, a particularly nasty type of skin cancer that responds poorly to conventional radiotherapy and chemotherapy, is sometimes treated with an immune-stimulating cytokine called interferon.
In some ways this century-old form of treatment is still a fringe area of medicine. But researchers have once again begun to probe the possibilities of immunotherapy. New antibody-based treatments like Mabthera and Herceptin are making a real difference in the treatment of common cancers like lymphoma and breast cancer. Although these therapies don’t stimulate the body’s immunity as a whole, they are based on antibody molecules which are key components of the human immune system. They show that our increasing knowledge of the molecular nitty-gritty of the body’s own defence and repair network is starting to make a real difference in the battle against cancer. One tumour at a time, such advances in modern medicine are finally vindicating William Coley and his one-hundred-year-old cancer-killing concoction.
Further reading:Written by Matt Castle on 13 July 2007
Coley Fluid
Coley Fluid is a modern reformulation of a cancer therapy
developed in 1893 by New York surgeon William Coley.
Over the years, numerous versions of Coley Fluid have been
made under different names including Coley’s Toxins, Coley
Vaccine, Coley’s Mixture, Erysipelas and Prodigiosis Toxins,
C-VAX, Febrivax, MBV (Multi Bacterial Vaccine), and
Vaccineurin. All of these products were sterile mixtures of
the bacteria Streptococcus pyogenes and Serratia marcescens,
but each of these products had a different specific formulation
and the limitations of historical quality control techniques
resulted in significant batch-to-batch variations in efficacy.
Certain formulations of Coley Fluid were much better than
others. The best version of Coley Fluid that was responsible
for the largest number of successful cases was Tracy Type XI
developed by Martha Tracy for the personal use of William
Coley and made between 1907 and 1922. MBVax Bioscience
Coley Fluid is equivalent to Tracy Type XI and the
company’s proprietary technology has reduced batch-to-
batch variations to a minimum.
Thousands of cancer patients were treated with Coley Fluid
during its more than 70 years as a mainstream cancer therapy.
The subsequent history of these patients shows Coley Fluid to
be as effective or more effective than modern therapies, and
in many cases curative.
A retrospective review was carried out on 896 histologically
confirmed cancers treated with Coley Fluid from 1893 up to
the early 1960s. For advanced inoperable tumors, a 50% or
greater 5-year survival was seen in multiple myeloma, giant
cell bone tumors, soft tissue sarcoma, lymphoma, melanoma,
breast, ovarian, cervical and uterine cancers (Nauts 1984)
In 1999, researchers studied 10-year survival of historical
Coley Fluid patients compared to 10-year survival of matched
controls from the National cancer Institute’s Surveillance
Epidemiology End Result data base (Alt Ther Health Med
1999; 5:42)
To support researchers wishing to conduct clinical trials,
MBVax Bioscience has developed a comprehensive Clinical
Protocol and Investigators Brochure and, providing
regulatory authorities properly authorize the clinical trial, the
company will supply this documentation and Coley Fluid
without charge.
==========================
MB Fluid
Bacterial substances and cytokines
MBVax Bioscience Inc. www.mbvax.com
Bacterial Substances
MB Fluid consists of a mixture of twotypes of killed bacteria: Streptococcus pyogenes is
Gram-positive (meaning a triphenylmethane dye called Gram stain is not decolorized by
ethanol) and Serratia marcescens is Gram-negative (the Gram stain is decolorized).
Gram-positive and Gram-negative bacteria contain different sets of substances that
stimulate the innate immune system:
* Gram-negative bacteria contain unmethylated CpG DNA sequences, the
endotoxin lipopolysaccharide (LPS), and peptidoglycan (PGN).
* Gram-positive bacteria alsocontain unmethylated CpG DNA sequences and
peptidoglycan (PGN), but donot contain LPS.
* Gram-positive bacteria alsocontain exotoxins, lipoteichoic acid (LTA),
streptolysin o(SLO) and cytoplasmic membrane-associated protein (CAP).
* In addition, both types of bacteria contain other, unidentified immune stimulatory
substances.
Bacterial DNA
Bacterial DNA contains unmethylated CpG sequences that bind tothe human Toll-like
receptor TLR9 (Bauer
1) and trigger an innate immune response leading tothe
secretion of IL-6, IL-10, IL-12, IP-10, TNF-a, IFN-a, IFN-ß and IFN-?.
2
Both CD4-
positive and CD4-negative peripheral blood dendritic precursor cells respond toCpG
DNA, but monocyte-derived DCs did not respond toCpG (Hartmann3).
Endotoxin (LPS)
LPS activates cells through the pattern recognition receptors CD14 and Toll-like
receptor 2 (TLR2) on monocytes, macrophages, endothelium and polymorphonuclear
neutrophils, thereby inducing the release of TNF-a, IL-6, and nitric oxide (Dziarski
4
,
Matsuura
5). Nitric oxide is cytostatic and/or cytolytic for tumor cells (Farias-Eisner
6).
LPS alsoinduces the production of IL-1a, IL-8, IL-10 (Bjork7), IP-10 (Luster
8), and
small quantities of TNF-ß (Hackett
9), and activates the complement pathway (Loos
10).
LPS is a B cell mitogen and polyclonal activator in mice (Dziarski
11). Monocyte-
derived DCs are highly sensitive toLPS, but both CD4-positive and CD4-negative
peripheral blood dendritic precursor cells show little response toLPS (Hartmann12).
3
Exotoxins (Spe)
Streptococcal pyrogenic exotoxins (Spe) are produced in the cell walls of
Streptococcus pyogenes and secreted intothe extracellular environment. Exotoxins
include SpeA, SpeB and SpeC, and a number of other exotoxins including SpeF,
SpeG, SpeZ, SSA, SMEZ and SMEZ-2 (Muller-Alouf
13). Exotoxins are both
pyrogenic (induces a fever) and mitogenic (induces cellular proliferation). Exotoxins
are pyrogenic because they stimulate the production of cytokines and chemokines.
Exotoxins are mitogenic because they function as “superantigens” which can give rise
topolyclonal activation (Marrack14
, Leonard15).
Superantigens have the ability tobind tomajor histocompatibility complex molecules
on antigen-presenting cells and simultaneously toT cell receptors, thereby triggering a
polyclonal expansion of T lymphocytes. The superantigen-mediated T cell activation
process has alsobeen shown toelicit a characteristic pattern of cytokines distinct from
that seen with LPS, including the T cell derived cytokines, IFN-? and IL-2 (Bjork16).
TNF-ß is induced more efficiently by the superantigens than by LPS (Hackett
17).
The best-characterized exotoxin, streptococcal pyrogenic exotoxin type A (SpeA),
stimulates the production of:
* Cytokines IL-1a, IL-6, TNF-a, IL-12, IL-10, IP-10;
* Th1 derived cytokines TNF-ß, IFN-?, IL-2;
* Th2 derived cytokine IL-5;
* IL-3, GM-CSF;
* Chemokines IL-8, RANTES and MIP-1-a (Muller-Alouf
18); and
* Enhances the host antibody response toother antigens (Hanna
19).
Peptidoglycan (PGN)
Peptidoglycan, a major component of the cell walls of Gram-positive bacteria, induces
the release of TNF-a (Dziarski
20) IL-8 (Wang21), IL-1 and IL-6 (Schwandner
22). PGN
is a B cell mitogen and polyclonal activator in mice (Dziarski
23). PGN is alsoa
constituent of the cell walls of Gram-negative bacteria.
Lipoteichoic acid (LTA)
Lipoteichoic acid alsobinds toCD14 (Dziarski, 1998), inducing release of TNF. LTA
induces TNF-a, IFN-a, IFN-ß and IFN-? in primed mice (Tsutsui
24); IL-1ß, IL-6 and
TNF in human monocyte cultures (Bhakdi
25
, Keller
26
, Yamamoto27); IL-8 and MIP-
1a (Gao28); and IL-12 (Cleveland29). LTA stimulates mitogenesis of T, but not B,
lymphocytes (Beachey30), and activates the complement pathway (Loos
31).
4
Streptolysin o(SLO)
Streptolysin O, produced by Streptococcus pyogenes, stimulates monocytes toproduce
IL-1ß and TNF-a (Hackett
32), and stimulates bone marrow-derived mast cells to
produce IL-4, IL-6, IL-13, GM-CSF, TNF-a and MCP-1 (Stassen33), and binds IgG
antibodies toform immune complexes with potent complement-activating capacity
(Bhakdi
34).
Cytoplasmic membrane-associated protein (CAP)
CAP is found in the cytoplasmic membrane but not in cell walls, peptidoglycan,
lipoteichoic acids, or cytoplasmic soluble fractions. This mitogenic factor produces
polyclonal activation of many classes of T lymphocytes (Itoh35).
Histone-like protein (HlpA)
HlpA is a constituent of Streptococcus pyogenes. Exposure of macrophages toa
mixture of HlpA and lipoteichoic acid resulted in a synergistic response in the
production of both TNF-a and IL-1 (Zhang36).
Dependent effects
The immune responses tobacterial substances are complex.
* Bacterial substances can synergistically enhance immune responses.
o TNF alone has a low systemic toxicity in tumor- and pathogen-free mice.
However, TNF given intravenously with nanogram quantities of LPS can
cause lethal shock (Rothstein37). Additional synergy might be expected to
occur from the presence of LPS and the streptococcal exotoxin itself
(Kim38), a similar combination of which has been recommended and
exploited as a method for the detection of LPS, the lethality of which was
found tobe enhanced by as much as 50,000-fold or more (Bohach39).
o Synergistic induction of TNF and IL-1 from macrophages has been
observed in vitrounder combined treatment with LPS and superantigens
(Parsonnet
40
, Beezhold41). In terms of IL-1ß production from human
monocytes, SpeA and Streptolysin otogether were synergistic: SpeA 193
pg/ml; Streptolysin O, 452 pg/ml; SpeA plus Streptolysin O, 799 pg/ml
(Hacket
42).
o Streptococcal pyrogenic exotoxins can enhance the host antibody response
toother antigens (Hanna
43).
5
* Bacterial substances can antagonistically reduce immune responses.
o Peptidoglycan-induced monokine production can be blocked by LPS
(Weidemann44).
* Bacterial substances can differentially induce proliferation of lymphocytes.
o Exotoxins include the classical Streptococcal pyrogenic exotoxins type A,
B and C, and a number of other exotoxins including type F, type G, type
Z, SSA, SMEZ and SMEZ-2. These exotoxins bind todifferent T cell
receptor motifs and thereby stimulate the expansion of different polyclonal
populations of T cells (Muller-Alouf
45).
o Lipoteichoic acid is alsomitogenic for T cells. Both T and B lymphocytes
possess a single population of specific binding sites of lipoteichoic acid,
and as a consequence of its binding, lipoteichoic acid stimulates
mitogenesis of T, but not B, lymphocytes (Beachey 46).
o Cytoplasmic membrane-associated protein (CAP) alsoproduces
polyclonal activation of many classes of T lymphocytes (Itoh 47).
* Bacterial substances can differentially induce the maturation of antigen-
presenting dendritic cells (DCs).
o Both CD4-positive and CD4-negative peripheral blood dendritic precursor
cells respond toCpG DNA, but these DCs showed little response toLPS.
In contrast, monocyte-derived DCs did not respond toCpG, but they were
highly sensitive toLPS (Hartmann 48).
* Bacterial substances induce the production of cytokines via different
pathways.
o The LPS receptor - CD14 - alsobinds lipoteichoic acid, inducing release
of TNF (Dziarski 49); but peptidoglycan (which alsoinduces TNF)
interacts via a different receptor because blocking CD14 had noinfluence
on Peptidoglycan induced TNF (Wang50). In mice, lipoteichoic acid
suppressed Meth A fibrosarcoma tumor growth and Peptidoglycan did not
- alsolipoteichoic acid induced TNF in Propionibacterium acnes-primed
mice, but Peptidoglycan did not (Usami 51).
* Bacterial substances induce the production of cytokines with different
kinetics.
o The kinetics of TNF-a production after stimulation is different for LPS,
Streptolysin oand SpeA. LPS immediately stimulates production, rising
toa max in 24 h then leveling off through 72 h. Production due toSpeA
and Streptolysin odoes not begin for 6 h, then rises following similar
6
patterns until 48 h, then SpeA continues torise while Streptolysin ofalls
and at 72 h is comparable toits level at 12 h (Hackett, 52 Fast 53).
Cytokines
The biological activity of MB Fluid can be described in terms of the cytokines and other
substances that mediate the immune response.
GM-CSF
Granulocyte-macrophage colony-stimulating-factor is a cytokine that stimulates
proliferation of granulocytes and macrophages, activates macrophages and promotes
the differentiation and maturation of dendritic cells. Activated T cells, macrophages,
endothelial cells and bone marrow stromal cells produce GM-CSF.
Oncolytic properties of GM-CSF
In the treatment of cancer, GM-CSF produced a 50% reduction in tumor volume in
a soft tissue sarcoma patient (Steward54). Injection of a murine tumorigenic T-
leukemia cell line expressing mGM-CSF intopre-established tumors of syngenic
mice led toa significant regression of these tumors (Hsieh55). Furthermore,
syngenic mice injected with melanoma cells or cells transfected with a recombinant
GM-CSF gene either completely rejected the tumor cells or developed tumors with
a mean volume fifty-times smaller than the control (Armstrong 56).
IL-1a, IL-1ß
There are twoforms of the cytokine interleukin-1, IL-1a and IL-1ß, coded by separate
genes and showing only 30% structural homology. Nevertheless, these twocytokines
bind the same receptors and have the same function: toinduce and promote
inflammatory reactions. IL-1 is produced by activated macrophages, and in smaller
quantities by neutrophils, epithelial cells (especially keratinocytes), and endothelial
cells.
Oncolytic properties of IL-1
When human IL-1ß was introduced intoB16 mouse melanoma cells, the growth of
B16 transfectants injected subcutaneously in syngenic mice was significantly
reduced (Bjorkdahl 57).
7
IL-2
The cytokine IL-2 is the major growth factor for antigen-activated T lymphocytes; it
alsopromotes B lymphocyte proliferation, antibody production, and activates NK
cells. IL-2 is produced by activated T lymphocytes, mostly CD4+
T cells and in smaller quantities by CD8+ T cells.
Oncolytic properties of IL-2
The FDA approved high-dose IL-2 for treatment of patients with metastatic kidney
cancer in 1992 and for metastatic melanoma in 1998 (Rosenberg58).
IL-3
IL-3 acts on immature bone marrow progenitors tostimulate the production of
lymphocytes. IL-3 is produced by CD4+ T lymphocytes.
IL-4
IL-4 participates in the activation of B-cells as well as other cell types. It is a co-
stimulator of DNA-synthesis, induces the expression of class II MHC molecules on
resting B-cells, and alsoenhances both secretion and cell surface expression of IgE
and IgG1. IL-4 alsostimulates cytotoxic T lymphocytes (CTLs). IL-4 is secreted by
Th1 cells (T helper cells, type 1).
Oncolytic properties of IL-4
IL-4 augments tumor immunogenicity and enhances the induction of tumor reactive
lymphoid cells in animal models (Krauss 59). Gene transfer of IL-4 intomouse
tumor cells has been shown tostimulate a strong immune response resulting in the
rejection of the transduced tumor when injected in vivo (Melani 60). Phase I/II
clinical trials have been conducted in which human autologous dermal fibroblasts
were cultured, transduced with the IL-4 gene, selected, irradiated, and administered
topatients as a vaccine (Elder 61).
IL-5
The cytokine IL-5 stimulates the growth and differentiation of eosinophils, activates
mature eosinophils, and stimulates the production of B lymphocytes and IgA
antibodies. The principal sources of IL-5 are the Th2 subset of activated CD4+
T lymphocytes and activated mast cells.
IL-6
IL-6 is a cytokine that plays a major role in inflammation, stimulates the synthesis of
acute phase proteins by hepatocytes, and serves as a growth factor for cells of the B-
cell lineage, especially terminally differentiated Ig-secreting plasma cells.
Mononuclear phagocytes, endothelial cells, fibroblasts, and other cells, in response to
bacterial substances and toother cytokines notably IL-1 and TNF, produce IL-6.
Oncolytic properties of IL-6
In SCID mice bearing human tumors and reconstituted with human CTL,
administration of a recombinant adenoviral vector expressing IL-6 induced human
CTL and inhibited growth and metastasis of the human tumor cells (Saggio62).
IL-8
IL-8 is a chemokine that attracts neutrophils, basophils, and T cells, but not
monocytes. It is alsoinvolved in neutrophil activation and is released from several cell
types in response toan inflammatory stimulus. Leukocytes and several types of tissue
cells produce IL-8.
Oncolytic properties of IL-8
Human IL-8 dramatically inhibited the tumor growth rate of CHocells in vivo
when injected intonude mice (Hirose
63).
IL-10
The cytokine IL-10 has potent anti-inflammatory properties. IL-10 is the major
inhibitor of activated macrophages. IL-10 inhibits the production of macrophage-
derived IFN-?, IL-2, IL-3, TNF and GM-CSF, thereby suppressing inflammation and
the Th1 pathway of T helper cell differentiation, and serving as negative feedback in
macrophage activation. IL-10 plays a role in adaptive immunity by enhancing the
proliferation of B lymphocytes. IL-10 is produced by activated macrophages.
Oncolytic properties of IL-10
Gene transfer studies have suggested that IL-10 induced tumor suppression is
mediated via enhanced natural killer (NK) cell activity (Gerard64
, Kundu65) as well
as inducible isoforms of nitric oxide synthase (Kundu66).
9
IL-12
The cytokine IL-12 is the principal mediator of early innate immune responses to
bacterial substances. The biological role of IL-12 is toinitiate a series of responses
involving macrophages, NK cells, and T lymphocytes. It is a potent stimulator of the
Th1 pathway of helper T cell differentiation, stimulates production of IFN-? by NK
cells and T lymphocytes, and enhances the cytolytic functions of activated NK cells
and CD8+
cytotoxic T lymphocytes (CTLs). The twoprincipal sources of IL-12 are
activated macrophages and dendritic cells.
Oncolytic properties of IL-12
The antitumor activity of IL-12 is documented by a large set of data from numerous
mouse models (Cavallo67). Gene transfer studies of IL-12 have been efficient at
reducing tumor growth and even complete eradication of established primary
tumors, as well as reduction of metastases in different tumor models (Hiscox68).
Also, IL-12 expression at the tumor site generated a long-term protective antitumor
immune response. IL-12 gene transfer is being tested in human clinical trials
(Sun 69).
IL-13
The cytokine IL-13 suppresses macrophage activation and antagonizes IFN-?. IL-13
alsoinduces the differentiation of dendritic cells. Th2 cells and some epithelial cells
produce IL-13.
Oncolytic properties of IL-13
IL-13 gene transfer induces anti-tumor protection due tothe stimulation of specific
antitumor effector cells (Lebel-Biany 70).
Interferon
IFN-a and IFN-ß, despite their structural differences, bind the same type I interferon
receptor and are therefore called type I interferon. IFN-a, sometimes called leukocyte
interferon, comprises a family of 20 species of molecules that are produced by a subset
of mononuclear phagocytes. IFN-ß, a single substance produced by a variety of cell
types, most notably fibroblasts, is alsocalled fibroblast interferon.
IFN-?, a single substance, is alsocalled immune interferon or type II interferon. It
exerts numerous biological effects including activating macrophages, enhancing the
expression of class I and class II MHC molecules, promoting the differentiation of
naïve CD4+
T cells tothe Th1 subset, inhibiting the proliferation of Th2 cells,
promoting the antibody class switch toIgG subclasses, inhibiting the class switch to
10
IgE, activating neutrophils, and enhancing the cytolytic activity of NK cells. NK cells,
CD4+
Th1 cells and CD8+ cells produce IFN-?.
Oncolytic properties interferon
IFN-a is an FDA approved treatment for hairy cell leukemia and melanoma, and is
being used as an investigational drug for numerous other cancers.
IFN-? induces macrophages torelease NO, which is cytostatic and/or cytolytic for
tumor cells (Farias-Eisner 71).
All types of interferon enhance the expression of MHC class I antigens and
promote the Th1 pathway of T helper differentiation by target cells, and induce
target cells todisplay the same class of immune epitopes as displayed by antigen
presenting cells such as dendritic cells, thereby allowing the detection and
destruction of tumor cells that might have otherwise been invisible tothe immune
system (Van den Eynde 72).
IP-10
Interferon-inducible protein-10 is a member of the chemokine family. IP-10 exerts a
chemotactic activity on lymphoid cells such as T cells, monocytes and NK cells. IP-10
is alsoa potent inhibitor of angiogenesis: it inhibits neovascularization by suppressing
endothelial cell differentiation. IP-10 is an IFN-? inducible protein that is produced
mainly by monocytes, but alsoby T cells, fibroblasts and endothelial cells.
Oncolytic properties of IP-10
Gene transfer of IP-10 intotumor cells reduced their tumorgenicity, and elicited a
long-term protective immune response (Luster
73). The angiostatic activity of IP-10
was shown tomediate tumor regression: tumor cells expressing IP-10 became
necrotic in vivo(Sgadari
74). IP-10 has alsobeen shown tomediate the angiostatic
effects of IL-12 that lead totumor regression (Tannenbaum 75).
MCP-1
Monocyte chemoattractant protein-1 (MCP-1) is a chemokine produced by a variety of
hematopoietic and non-hematopoietic cell types. MCP-1 attracts monocytes, T and NK
cells.
Oncolytic properties of MCP-1
Gene transfer of MCP-1 intotumor cells demonstrated antitumor effects
(Manome 76).
11
MIP-1a
Macrophage inflammatory protein-1a is a chemokine. MIP-1a attracts monocytes,
neutrophils, eosinophils, dendritic cells, NK, and T cells.
Oncolytic properties of MIP-1a
MIP-1a exerts an antitumoral effect because of its ability torecruit immune cells at
the tumor site. In mice, MIP-1a elicited a long-term immune response that resulted
in protection of the animals against challenge by tumor cells (Nakashima
77).
RANTES
RANTES is a chemokine that attracts monocytes, dendritic, T and NK cells,
eosinophils and basophils.
Oncolytic properties of RANTES
Tumor cells transduced with the RANTES gene had a reduced ability toform
tumors in vivo, and elicited an anti-tumor immune response that protected animals
from challenge with the parent tumor cells (Mule 78).
TNF-a, TNF-ß
Tumor necrosis factor alpha is a cytokine that induces and promotes inflammatory
reactions involving recruitment of neutrophils and monocytes tothe site of infection,
and activation of these cells. Additionally, TNF-a stimulates endothelial cells, and also
macrophages, tosecrete chemokines that further increases the migration of leukocytes
from blood totissue. TNF-a alsostimulates the secretion of IL-1 by macrophages.
TNF alsoenhances the antibody response. LPS-activated macrophages, antigen-
activated T lymphocytes, NK cells and mast cells produce TNF-a. The target of TNF-
a is any cell (all human cell types express TNF receptors).
Tumor necrosis factor beta is similar in biological effect and structure toTNF (but it is
a different molecule). Alsocalled Lymphotoxin (LT), TNF-ß is produced by some
antigen-activated T lymphocytes in smaller quantities than the TNF-a made by
macrophages, therefore TNF-ß does not exert systemic effects but acts like a local
promoter of inflammation. TNF-ß is induced more efficiently by the superantigens
than by LPS (Hackett
79).
12
Oncolytic properties of TNF
As its name implies, TNF has the ability todestroy tumors. Researchers have
achieved 90% complete response rates by employment of isolated limb perfusion to
deliver high local concentrations of TNF toselected patients with melanoma and
sarcoma (Lienard80). TNF has been shown tofacilitate the in vivolocalization of
radiolabelled monoclonal antibodies at the site of the tumor towhich they were
directed (Smyth 81).
13
Bacterial Substances and Immune Mediators
Serratia marcescens
Streptococcus pyogenes
CpG
LPS
CpG
Spe
PGN
LTA
SLO
CAP
GM-CSF X X
IL-1 X X X X X
IL-2 X
IL-3 X
IL-4 X
IL-5 X
IL-6 X X X X X X X
IL-8 X X X X
IL-10 X X X X
IL-12 X X X X
IL-13 X
IFN-a X X X
IFN-ß X X X
IFN-? X X X X
IP-10 X X X X
MCP-1 X
MIP-1a X X
RANTES X
TNF-a X X X X X X X
TNF-ß X X
Inducer of mitogenesis, enhancement or maturation of:
T lymphocytes X X X
B lymphocytes X X
Dendritic cells X X X
Complement X X X
14
References
1
Bauer S, Kirschning CJ, Hacker H, et al. Human TLR9 confers responsiveness tobacterial DNA via
species-specific CpG motif recognition. PNAS 2001; 98(16):9237.
2
“CpG mechanism of action” www.coleypharma.com/wt/coley/cpg_action Accessed Dec 17, 2003.
3
Hartmann G, Weiner GJ, Krieg AM. CpG DNA: a potent signal for growth, activation, and maturation of
human dendritic cells. PNAS 1999; 96:9305.
4
Dziarski R, Tapping R, Tobias PS. Binding of bacterial peptidoglycan toCD14. J Biol Chem 1998;
273:8680.
5
Matsuura M, KisoM, Hasegawa A. Activity of monosaccharide lipid A analogues in human monocytic
cells as agonists or antagonists of bacterial lipopolysaccharide. Inf and Immunity 1999; 67(12):6286.
6
Farias-Eisner R, Sherman MP, Aeberhard E, Chaudhuri G. Nitric oxide is an important mediator for
tumoricidal activity in vivo. PNAS 1994; 91:9407.
7
Bjork L, et al. Endotoxin and Staphylococcus aureus enterotoxin A induce different patterns of cytokines.
Cytokine 1992; 4:513.
8
Luster, A.D., and Leder, P. (1996) IP-10, a CXC chemokine, elicits a potent thymus-dependent antitumor
response in vivo. J. Exp. Med. 178: 1057-1065.
9
Hackett SP, Stevens DL. Superantigens associated with staphylococcal and streptococcal toxic shock
syndrome are potent inducers of tumor necrosis factor-beta synthesis. J Infect Dis 1993; 168:232.
10
Loos M, Clas F, Fischer W. Interaction of purified lipoteichoic acid with the classical complement
pathway. Inf Immunity 1986; 53(3):595.
11
Dziarski R. Studies on the mechanism of peptidoglycan- and lipopolysaccharide-induced polyclonal
activation. Inf Immunity 1982; 35(2):507.
12
Hartmann G, Weiner GJ, Krieg AM. CpG DNA: a potent signal for growth, activation, and maturation of
human dendritic cells. PNAS 1999; 96:9305.
13
Muller-Alouf H, Proft T, Zollner TM, et al. Pyrogenicity and cytokine-inducing properties of
Streptococcus pyogenes superantigens: comparative study of streptococcal mitogenic exotoxin Z and
pyrogenic exotoxin A. Inf and Immunity 2001; 69(6):4141.
14
Marrack P, Kappler J. The staphylococcal enterotoxins and their relatives. Science 1990; 248:705.
15
Leonard BA, et al. Cell and receptor requirements for streptococcal pyrogenic exotoxin T cell
mitogenicity. Infect Immunity 1991; 59:1210.
16
Bjork L, et al. Endotoxin and Staphylococcus aureus enterotoxin A induce different patterns of
cytokines. Cytokine 1992; 4:513.
17
Hackett SP, Stevens DL. Superantigens associated with staphylococcal and streptococcal toxic shock
syndrome are potent inducers of tumor necrosis factor-beta synthesis. J Infect Dis 1993; 168:232.
18
Muller-Alouf H, Proft T, Zollner TM, et al. Pyrogenicity and cytokine-inducing properties of
Streptococcus pyogenes superantigens: comparative study of streptococcal mitogenic exotoxin Z and
pyrogenic exotoxin A. Inf and Immunity 2001; 69(6):4141.
15
19
Hanna EE, Watson DW. Enhanced immune response after immunosuppression by streptococcal
pyrogenic exotoxin. Infect Immun 1973; 7:1009.
20
Dziarski R, Tapping R, Tobias PS. Binding of bacterial peptidoglycan toCD14. J Biol Chem 1998;
273:8680.
21
Wang Q, Dziarski R, Kirschning CJ, et al. Micrococci and peptidoglycan activate
TLR2>MyD88>IRAK>TRAF>NIK>IKK>NF-kappa-B signal transduction pathway that induces
transcription of interleukin-8. Inf Immunity 2001; 69(4):2270.
22
Schwandner R, Dziarski R, Wesche H, et al. Peptidoglycan- and lipoteichoic acid-induced cell activation
is mediated by toll-like receptor 2. J Biol Chem 1999; 274(25):17406.
23
Dziarski R. Studies on the mechanism of peptidoglycan- and lipopolysaccharide-induced polyclonal
activation. Inf Immunity 1982; 35(2):507.
24
Tsutsui O, Kokeguchi S, Matsumura T, KatoK. Relationship of the chemical structure and
immunobiological activities of lipoteichoic acid from Streptococcus faecalis (Enterococcus hirae) ATCC
9790. FEMS Microbiol Immunol 1991; 76:211.
25
Bhakdi S, Klonisch T, Nuber P, Fischer W. Stimulation of monokine production by lipoteichoic acids.
Infect Immun 1991; 59:4614.
26
Keller R, Fischer W, Keist R, Bassetti S. Macrophage response tobacteria: induction of marked secretory
and cellular activities by lipoteichoic acids. Infect Immunity 1992; 60:3664.
27
YamamotoA, Usami H, Nagamuta M, et al. The use of lipoteichoic acid (LTA) from Streptococcus
pyogenes toinduce a serum factor causing tumor necrosis. Br J Cancer 1985; 51:739.
28
GaoJJ, Xue Q, Zuvanich EG, et al. Commercial preparation of lipoteichoic acid contain endotoxin that
contributes toactivation of mouse macrophages in vitro. Inf Immunity 2001; 69(2):751
29
Cleveland MG, Gorham JD, Murphy TL, et al. Lipoteichoic acid preparations of gram-positive bacteria
induce interleukin-12 through a CD14-dependent pathway. Inf Immunity 1996; 64(6):1906.
30
Beachey EH, et al. Lymphocyte binding and T cell mitogenic properties of group A streptococcal
lipoteichoic acid. J Immunol 1979; 122:189.
31
Loos M, Clas F, Fischer W. Interaction of purified lipoteichoic acid with the classical complement
pathway. Inf Immunity 1986; 53(3):595.
32
Hackett SP, Stevens DL. Streptococcal toxic shock syndrome: synthesis of tumor necrosis factor and
interleukin-1 by monocytes stimulated with pyrogenic exotoxin A and streptolysin O. J Infect Dis 1992;
165:879.
33
Stassen M, Muller C, Richter C, et al. The streptococcal exotoxin streptolysin oactivates mast cells to
produce tumor necrosis factor alpha by p38 mitogen-activated protein kinase- and protein kinase C-
dependent pathways. Inf Immunity 2003; 71(11):6171.
34
Bhakdi S, Tranum-Jensen J. Complement activations and attack on autologous cell membranes induced
by streptolysin O. Inf Immunity 1985; 48(3):713.
16
35
Itoh T, Satoh H, IsonoN, et al. Mechanism of stimulation of T cells by Streptococcus pyogenes: isolation
of a major mitogenic factor, cytoplasmic membrane-associated protein. Inf and Immunity 1992;
60(8):3128.
36
Zhang L, Ignatowski TA, Spengler RN, et al. Streptococcal histone induces murine macrophages to
product inteleukin-1 and tumor necrosis factor alpha. Inf Immunity 1999; 67(12):6473.
37
Rothstein JL, Schreiber H. Synergy between tumor necrosis factor and bacterial products causes
hemorrhagic necrosis and lethal shock in normal mice. PNAS 1988; 85:607.
38
Kim YB, Watson DW. A purified group A streptococcal pyrogenic exotoxin - physicochemical and
biological properties including the enhancement of susceptibility toendotoxin lethal shock. J Exp Med
1970; 131:611.
39
Bohach GA, Schlievert PM. Detection of endotoxin by enhancement with toxic shock syndrome toxin-1
(TSST-1). Meth Enzymol 1988; 165:302.
40
Parsonnet J, Gillis ZA. Production of tumor necrosis factor by human monocytes in response totoxic-
shock-syndrome toxin-1. J Infect Disease 1988; 158:1026.
41
Beezhold DH et al, Synergistic induction of interleukin-1 by endotoxin and toxic shock syndrome toxin-1
using rat macrophages. Infect Immunity 1987; 55:2865.
42
Hackett SP, Stevens DL. Streptococcal toxic shock syndrome: synthesis of tumor necrosis factor and
interleukin-1 by monocytes stimulated with pyrogenic exotoxin A and streptolysin O. J Infect Dis 1992;
165:879.
43
Hanna EE, Watson DW. Enhanced immune response after immunosuppression by streptococcal
pyrogenic exotoxin. Infect Immun 1973; 7:1009.
44
Weidemann B, Brade H, Rietschel ET, et al. Soluble peptidoglycan-induced monokine production can be
blocked by anti-CD14 monoclonal antibodies and by lipid A parital structures. Inf Immunity 1994;
62(11):4709.
45
Muller-Alouf H, Proft T, Zollner TM, et al. Pyrogenicity and cytokine-inducing properties of
Streptococcus pyogenes superantigens: comparative study of streptococcal mitogenic exotoxin Z and
pyrogenic exotoxin A. Inf and Immunity 2001; 69(6):4141.
46
Beachey EH, et al. Lymphocyte binding and T cell mitogenic properties of group A streptococcal
lipoteichoic acid. J Immunol 1979; 122:189.
47
Itoh T, Satoh H, IsonoN, et al. Mechanism of stimulation of T cells by Streptococcus pyogenes: isolation
of a major mitogenic factor, cytoplasmic membrane-associated protein. Inf and Immunity 1992;
60(8):3128.
48
Hartmann G, Weiner GJ, Krieg AM. CpG DNA: a potent signal for growth, activation, and maturation of
human dendritic cells. PNAS 1999; 96:9305.
49
Dziarski R, Tapping R, Tobias PS. Binding of bacterial peptidoglycan toCD14. J Biol Chem 1998;
273:8680.
50
Wang JE, Jorgensen PF, Almlof M, et al. Peptidoglycan and lipoteichoic acid from Staphylococcus
aureus induce tumor necrosis factor alpha, inteleukin 6 (IL-6), and IL-10 production in both T cells and
monocytes in a human whole bllod model. Inf and Immunity 2000; 68(7):3965.
17
51
Usami H, et al. Antitumor effects of streptococcal lipoteichoic acids on Meth A fibrosarcoma. Br J
Cancer 1988; 57:70.
52
Hackett SP, Stevens DL. Streptococcal toxic shock syndrome: synthesis of tumor necrosis factor and
interleukin-1 by monocytes stimulated with pyrogenic exotoxin A and streptolysin O. J Infect Dis 1992;
165:879.
53
Fast DJ, et al. Toxic shock syndrome-associated staphylococcal and streptococcal pyrogenic toxins are
potent inducers of tumor necrosis factor production. Infect Immunity 1989; 57:291.
54
Steward WP, et al. Recombinant human granulocyte macrophage colony stimulating factor (rhGM-CSF)
given as daily short infusions - a phase I dose-toxicity study. Br J Cancer 1989; 59:142.
55
Hsieh CL, Pang VF, Chen DS, Hwang LH (1997). Regression of established mouse leukemia by GM-
CSF-transduced tumor vaccine: implications for cytotoxic T lymphocyte responses and tumor burdens.
Hum Gene Ther 1997 Nov 1;8(16):1843-1854
56
Armstrong, C. A., Botella, R., Galloway, T. H., Murray, N., Kramp, J. M., Song, I. S., and Ansel, J. C.
(1996). Antitumor effects of granulocyte-macrophage colony-stimulating factor production by melanoma
cells. Cancer Res. 56: 2191-2198.
57
Bjorkdahl, O., Wingren, A. G., Hedhund, G., Ohlsson, L., and Dohlsten, M. (1997) Gene transfer of a
hybrid interleukin-1 beta gene toB16 mouse melanoma recruits leucocyte subsets and reduces tumour
growth in vivo. Cancer Immunol. Immunother. (1997) 44: 273-281
58
Rosenberg SA. Progress in the development of immunotherapy for the treatment of patients with cancer.
J Intern Med 2001; 250(6):462-75.
59
Krauss, J. C., Cameron, M. J., Park, A. N., Forslund, K., and Chang, A. E. (1995). Efficient transduction
of early passage human melanoma tosecrete IL-4. J. Immunol. Methods 183: 239-250.
60
Melani, C., Chiodoni, C., Arienti, F., Maccalli, C., Sule-Suso, J., Anichini, A., Colombo, M. P., and
Parmiani, G. (1994). Cytokine gene transduction in tumor cells: interleukin (IL)-2 or IL-4 gene transfer in
human melanoma cells. Nat. Immun. 13: 76-84.
61
Elder, E. M., Lotze, M. T., and Whiteside, T. L. (1996). Successful culture and selection of cytokine
gene-modified human dermal fibroblasts for the biologic therapy of pateints with cancer. Hum. Gene. Ther.
7: 479-487.
62
SaggioI, Ciapponi L, SavinoR, et al. Adenovirus-mediated gene transfer of a human IL-6 antagonist.
Gene Ther. 4: 839-845.
63
Hirose K, Hakozaki M, Nyunoya Y, et al. Chemokine gene transfection intotumour cells reduced
tumorigenicity in nude mice in association with neutrophilic infiltration. Br. J. Cancer 1995; 72: 708-714.
64
Gerard, CM, Bruyns C, Delvaux A, et al. Loss of tumorigenicity and increased immunogenicity induced
by interleukin-10 gene transfer in B16 melanoma cells. Hum. Gene. Ther 1996; 7: 23-31.
65
Kundu, N., and Fulton, A. M. (1997). Interleukin-10 inhibits tumor metastasis, downregulates MHC class
I, and enhances NK lysis. Cell. Immunol. 180: 55-61.
66
Kundu N, Dorsey R, Jackson MJ, Guiterrez P, Wilson K, Fu S, Ramanujam K, Thomas E, Fulton AM
(1998) Interleukin-10 gene transfer inhibits murine mammary tumors and elevates nitric oxide. Int J Cancer
76(5):713-9
18
67
CavalloF, Di CarloE, Butera M, et al. Immune events associated with the cure of established tumors and
spontaneous metastases by local and systemic IL-12. Cancer Res 1999; 59:414-21.
68
Hiscox, S. and Jiang, W.G. (1997). Interleukin-12, an emerging anti-tumour cytokine. In Vivo11: 125-
132.
69
Sun, Y., et al. (1998). Vaccination with IL-12 gene-modified autologous melanoma cells: preclinical
results and first clinical phase I study. Gene Ther. 5: 481-490.
70
Lebel-Biany et al. 1995. Experimental gene therapy of cancer using tumor cells engineered tosecrete
interleukin-13. Eur. J. Immunol. 25: 2340-2348.
71
Farias-Eisner R, Sherman MP, Aeberhard E, Chaudhuri G. Nitric oxide is an important mediator for
tumoricidal activity in vivo. PNAS 1994; 91:9407.
72
Van den Eynde BJ, Morel S. Differential processing of class-I-restricted epitopes by the standard
proteasome and the immunoproteasome. Current Opinion in Immunol 2001; 13:147.
73
Luster, A.D., and Leder, P. (1996) IP-10, a CXC chemokine, elicits a potent thymus-dependent antitumor
response in vivo. J. Exp. Med. 178: 1057-1065
74
Sgadari, S., et al. (1996) Interferon-inducible protein-10 identified as a mediator of tumor necrosis in
vivo. Proc. Natl. Acad. Sci. USA 93: 13791-13796
75
Tannenbaum, C.S., Raymond, T., Armstrong, D., Finke, J.H., Bukowski, R.M., and Hamilton, T.A.
(1998) The CXC chemokines IP-10 and Mig are necessary for IL-12-mediated regression of the mouse
RENCA tumor. J. Immunol. 161: 927-932
76
Manome et al. 1995. Monocyte chemoattractant protein-1 (MCP-1) gene transduction: an effective tumor
vaccine strategy for non-cranial tumors. Cancer Immunol. Immunother. 41: 227-235.
77
Nakashima, E., Oya, A., Kubota, Y., et al. A candidate for cancer gene therapy: MIP-1 alpha gene
transfer toan adenocarcinoma cell line reduced tumorigenicity and induced protective immunity in
immunocompetent mice. Pharm Res 1996; 13: 1896-1901.
78
Mule JJ, Custer M., Averbook B, et al. RANTES secretion by gene-modified tumor cells results in loss of
tumorigenicity in vivo: role of immune cell subpopulations. Hum Gene Ther 1996; 7: 1545-1553.
79
Hackett SP, Stevens DL. Superantigens associated with staphylococcal and streptococcal toxic shock
syndrome are potent inducers of tumor necrosis factor-beta synthesis. J Infect Dis 1993; 168:232.
80
Lienard D, EwalenkoP, Delmotte JJ, et al. High does recombinant tumor necrosis factor alpha in
combination with interferon gamma and melphalan in isolation perfusion of the limbs for melanoma and
sarcoma. J Clin Oncol 1992; 10:52-60.
81
Smyth MJ, Pietersz GA, McKenzie IFC. Increased antitumor effect of immunoconjugates and tumor
necrosis factor in vivo. Cancer Res 1988; 48:3607-12.
== LA TIMES ARTICLE ==
Germs may actually help our bodies fight tumors - which
means an infection-free lifestyle comes at a price
From Los Angeles Times
IN the 1890s, a New York surgeon named William Coley
tested a radical cancer treatment. He took a hypodermic
needle teeming with bacteria and plunged it into the flesh
of patients.
After suffering through weeks of chills and fevers, many
showed significant regression of their tumors, but even
Coley himself could not explain the phenomenon.
His experiments were sparked by the observation that
certain cancer patients improved after contracting
infections. One patient experienced regression in a tumor
in her arm after developing Saint Anthony’s fire, a
streptococcus skin infection.
Doctors at the time considered Coley’s bacterial mixtures
to be more black magic than medicine, and with the advent
of radiation therapy, the well-meaning doctor was soon
consigned to the annals of quackery.
But today, some scientists think Coley had it right: Germs
can teach our bodies how to fight back against tumors. Dr.
John Timmerman, a cancer immunotherapy expert at UCLA’s
Jonsson Cancer Center, says this revolution has produced
"the most exciting sets of compounds in cancer
immunology."
These scientists have not yet proved their case. But new
studies are revealing that certain cancers may be reduced
by exposure to disease-causing bacteria and viruses, and
pharmaceutical companies are testing anticancer treatments
that capitalize on the concept by using bacterial elements
to boost the body’s natural immunity.
The studies also imply that our cleaner, infection-free
lifestyles may be contributing to the rise in certain
cancers over the last 50 years, scientists say, because
they make the immune system weaker or less mature. Germs
cause disease but may also fortify the body, a notion
summed up in a 2006 report by a team of Canadian
researchers as "whatever does not kill me makes me
stronger."
Almost a century after Coley, in the 1980s, dermatologists
began noticing that patients with severe acne, which is
caused by another type of bacterium, have reduced rates of
skin cancer, lymphoma and leukemia. According to a paper
by Dr. Mohammad Namazi at the Shiraz University of Medical
Sciences in Iran, studies showed that these bacteria, when
injected into animals, appear to stimulate the immune
system and shrink tumors.
More recent evidence for this phenomenon comes from
studies on cotton and livestock workers, who are
constantly breathing endotoxins, a component of bacterial
cell walls that causes swelling of lung tissue.
In reports published in the last two years, Harvey
Checkoway, a University of Washington epidemiologist, has
found that female cotton workers in Shanghai have a 40% to
60% lower risk of lung, breast, and pancreas cancer than
other factory workers.
Other recent studies by Giuseppe Mastrangelo at the
University of Padua in Italy found that dairy farmers
exposed to high levels of manure dust are up to five times
less likely to develop lung cancer than their colleagues
who work in open fields.
For the dairy farmers and cotton workers, "it’s good news
and bad news," Checkoway says. They have lower rates of
cancer but tend to have higher rates of other respiratory
problems. Sniffing cotton dust or inducing pimples is
never going to be a therapy, he says, but studying the
body’s reactions to bacteria could explain why cancer
rates go down upon endotoxin exposure. And that might help
in developing anticancer drugs.
Dr. Arthur Krieg, chief scientific officer of the
Boston-based Coley Pharmaceutical Group, thinks the
success of Coley’s toxins comes largely from a difference
between DNA of humans and bacteria and viruses.
In 1995, Krieg was at the University of Iowa working with
strands of DNA created in the lab, hoping to find a way to
turn off genes involved in the autoimmune disease lupus.
To his surprise, this DNA stimulated the immune cells he
was studying in lab dishes. "I got interested, and I got
puzzled," he says.
His synthetic DNA contained several regions called CpGs.
In humans, that region has a kind of chemical "cap" on it,
but bacteria - and Krieg’s synthetic DNA - lack that cap.
Thus, in effect, exposure to that CpG makes the body
"think" it’s being assaulted by pathogens, and triggers
the immune system to shift into attack mode - and, in
doing so, more effectively battle cancer cells.
Krieg saw medical potential: Maybe one could design small
drugs with CpGs in them and use them as immunity boosters.
After patenting the method, he left his university job and
founded Coley Pharmaceutical Group, which was acquired by
the New York-based drug company Pfizer in January.
Five years after his discovery, Krieg’s first compound has
proved safe in early trials but has not yet been proved
effective. Last year, the injected compound failed to
increase survival time in a trial of 1,600 lung cancer
patients also undergoing chemotherapy. But Krieg thinks it
will prove effective in other patients: "It’s just a
matter of finding the right way to use it," he says.
Timmerman is a strong believer in CpGs, and has been using
them with the antibody drug Rituximab in his lab research
on mice. Finding the right drug combination is key, he
says: "It’s very naive to think that a single
off-the-shelf immune stimulant is going to magically treat
cancer."
Krieg’s CpG-based chemicals have proved useful in another
arena. Because CpGs boost the immune system, they also can
enhance certain vaccines. In a trial sponsored by the U.S.
Defense Advanced Research Projects Agency, one of Krieg’s
chemicals, VaxImmune, accelerated the body’s response to
anthrax vaccine by a factor of two - from 40 days to 20.
Other collaborations are exploring using CpG-containing
DNA to develop hepatitis B vaccines and anti-asthmatic
drugs. (The company has many CpG compounds, four in
clinical trials.)
But Don MacAdam, chief executive of MBVax Bioscience in
Ancaster, Canada, is not sure that the healing properties
of Coley’s fluids are due to a short strand of DNA. "The
immune system is very complicated," he says, "Any of these
therapies that are doing one little thing are very likely
to fail."
And so MacAdam wants to revive the formulation that Coley
himself found most effective - a mixture of two kinds of
bacteria, Streptococcus and Serratia. Such an extract
would contain naturally occurring CpGs, endotoxins and
other bacterial components that may have therapeutic
potential.
MacAdam has solved Coley’s major difficulty: maintaining
consistency of the brew from batch to batch. His
preparation has been tested on terminal cancer patients
outside the U.S. and Canada, and he contends that 24 in 38
patients have shown signs of tumor regression, although
nothing is published yet. Dr. Vikas Sukhatme, a professor
at Harvard Medical School, says he hopes to run clinical
trials once the product has been manufactured according to
Food and Drug Administration guidelines.
Other groups have been experimenting with injections of
other types of heat-killed bacteria, including
Myobacterium vaccae, a tuberculosis relative. In two
studies in January’s European Journal of Cancer,
researchers report that these bacteria may help fight
certain lung and renal cancers.
The first study is a reanalysis of a trial with 162
patients who received heat-killed bacteria (and
chemotherapy). In the original study, the treatment didn’t
seem to improve survival and in 2004, the company
developing the therapy, London-based Silence Therapeutics,
gave up on Mycobacterium.
But John Stanford, a shareholder in the company and a
researcher at University College London, says these
studies were poorly designed and analyzed. When he and
collaborators re-analyzed the results, they found that
Mycobacterium injections could increase survival of
adenocarcinoma patients by four months. Stanford believes
that part of the bacterial cell wall switches the body
from producing ineffective antibodies to sending out
cancer-killing blood cells.
In the second study, researchers reported that 60 renal
cancer patients injected with Myobacterium survived just
as long as those treated with standard chemotherapy.
Stanford has formed a company, Immodulon Therapeutics, and
wants to run trials with a stricter and more intense
injection regime, to repeat the results and, hopefully,
extend patients’ lives longer.
Although both Krieg and Timmerman are inspired by Coley’s
work, they question the philosophy behind reviving Coley’s
preparation and using other bacterial extracts. But, Krieg
says, "as a physician you have to maintain a sense of
humility and avoid being overly skeptical."
No comments:
Post a Comment